
Veronica Ciocanel

Veronica Ciocanel
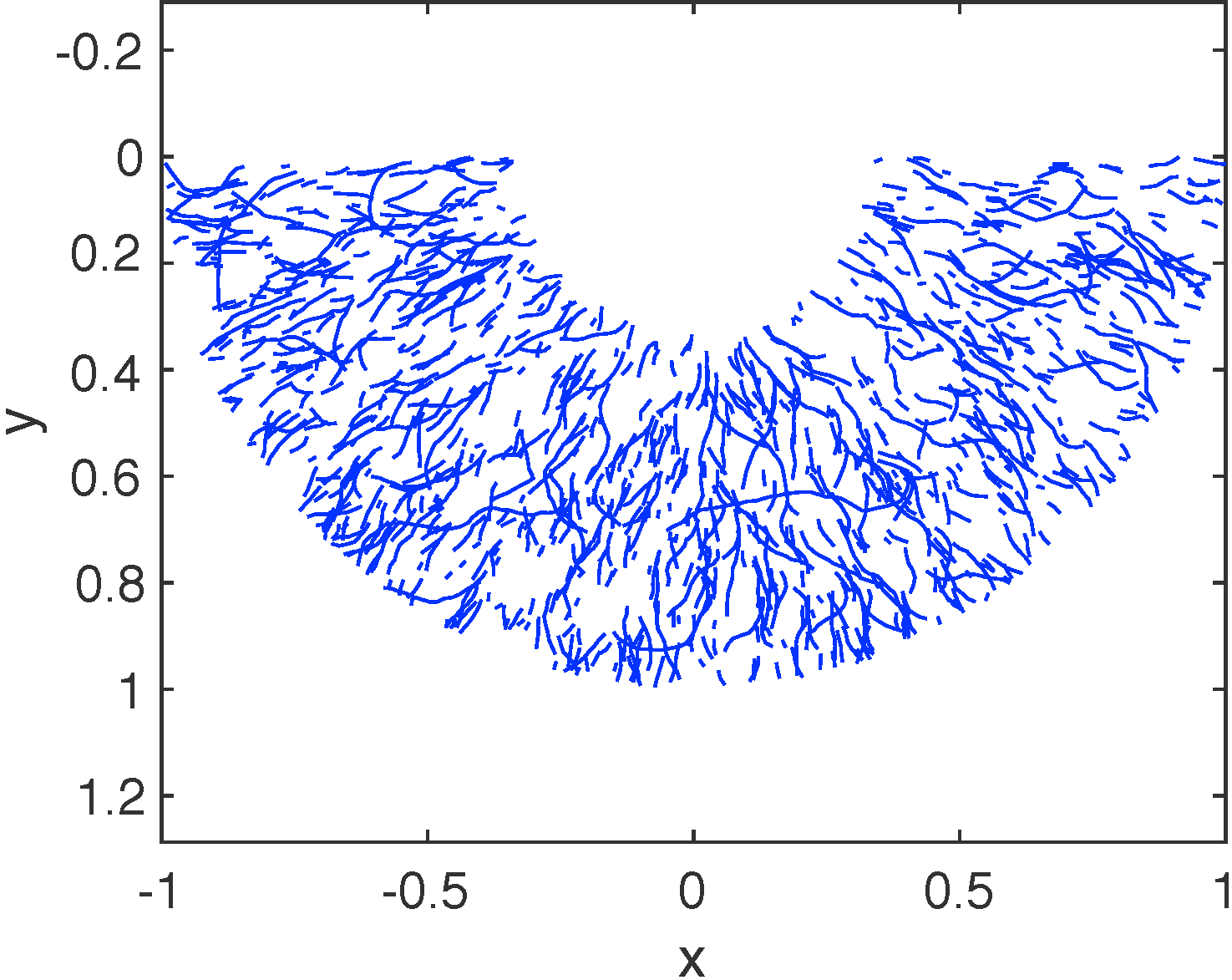
I am interested in understanding spatial differentiation in egg cells, which is critical in early development of organisms. Oocytes of the frog develop a spatial axis through localization of mRNA. I collaborate with the Mowry Lab and with Bjorn Sandstede at Brown University to study protein accumulation at the cell periphery (mRNA localization) using systems of advection-reaction-dffusion PDEs. We have developed novel methods to estimate transport parameters using fluorescence microscopy (FRAP) data. We also use dynamical systems analysis to predict large-time dynamics of mRNA particles. Recent work includes analytical and simulation work for mRNA dynamics with transport restricted to different microtubule (cellular roads) geometries.
Collaborators: Björn Sandstede (Brown University), Kimberly Mowry Lab (Brown University).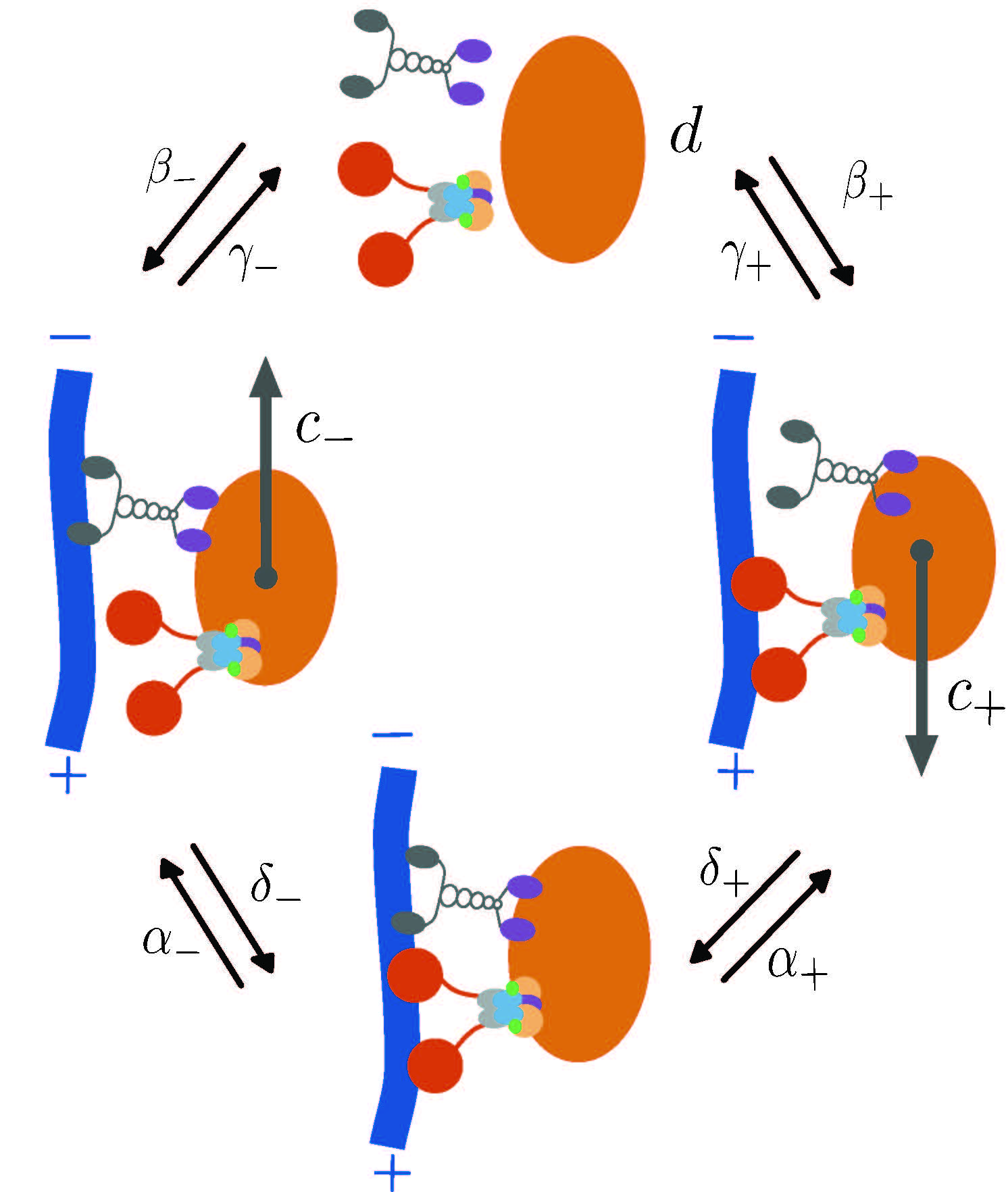
Intracellular particles often switch between different biophysical states such as active transport, diffusive transport, and paused states. Stochastic methods provide intuitive models for bridging between individual transport parameters and effective (large-time) transport properties. We use a renewal reward framework to study the dynamics of general transport models and illustrate their application to several models of cellular cargo movement.
Collaborators: Scott McKinley (Tulane University), John Fricks (ASU), Peter Kramer (RPI).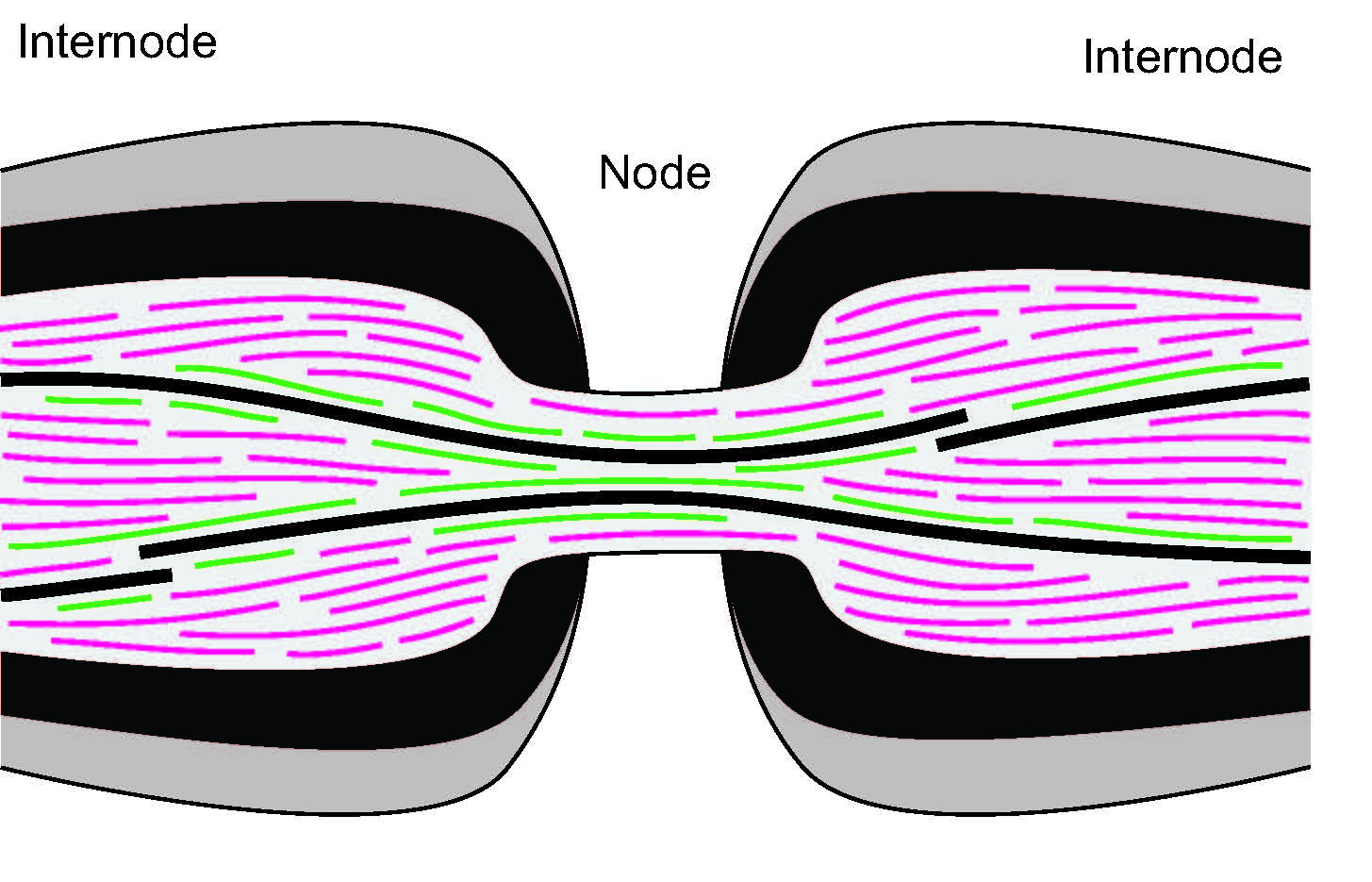
In axons of neurons, the transport of proteins called neurofilaments is key in ensuring a proper speed of neuronal communication. Neurofilaments have to move through constrictions called nodes of Ranvier, and defects in this transport are associated with a range of neurodegenerative diseases. We use an experimentally-backed stochastic model of bidirectional transport to investigate mechanisms that prevent bottlenecks at these constrictions. Improved access to microtubule tracks in the nodes may explain the healthy passage and distribution of neurofilaments.
Collaborators: Tony Brown (OSU), Peter Jung (Ohio University).In the worm C. elegans, contractile ring channels made of actin filaments allow for nutrient exchange to developing egg cells. We are developing agent-based models to understand how two myosin motor proteisn exert forces on these filaments to maintain the contractile rings at a constant diameter. In the video, actin filaments are in red, myosin motors are in blue, and cross-linkers are in black. We are also using in vitro experiments of myosin motors from collaborators to connect these models with several data analytic measurements.
Collaborators: Adriana Dawes (OSU), David Altman (Willamette University), Tim Atherton (Tufts University), Papoian Lab (University of Maryland).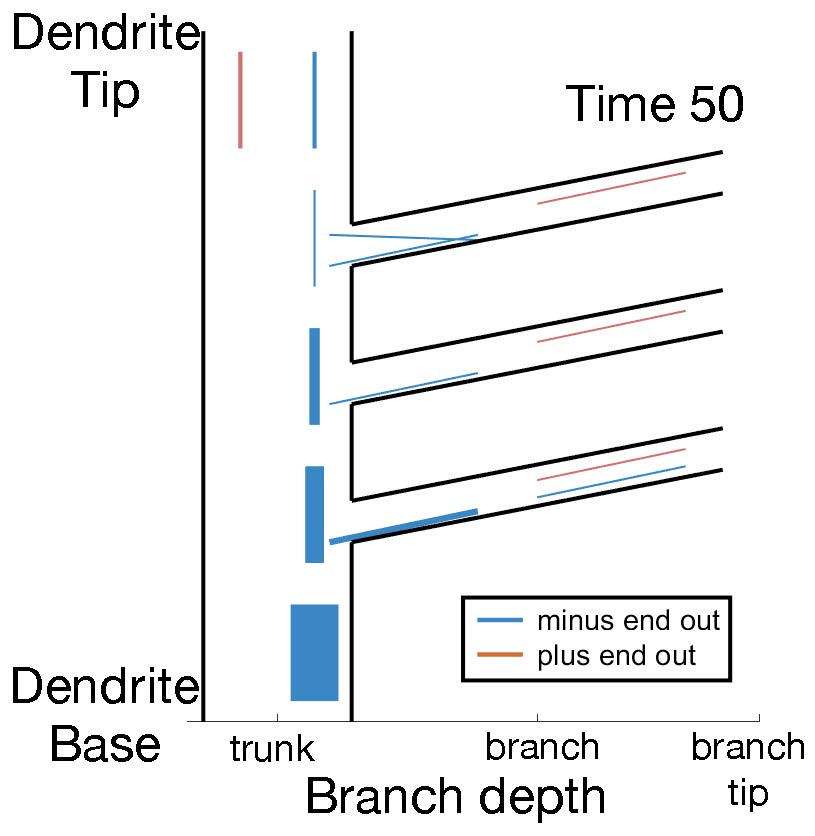
In neurons, dendrites have different functions over lifetime. The microtubule organization inside dendrites provides tracks for long-distance transport of many proteins and mRNAs. In the Drosophila fruit fly, neurons have highly branched dendrites where microtubules are consistently organized with their minus ends out (blue lines in the video). I am studying mechanisms that control microtubule orientations in dendrites using stochastic modeling. We focus both on maintenance and development of this polarity in dendrites, and are extending our approach to predict cytoskeleton reorganization during axonal injury.
Collaborators: Melissa Rolls Lab (Penn State University), Scott McKinley (Tulane University).